Cowden syndrome
This material supplements information provided by physicians, nurses, genetic counselors, and other members of your health care team about Cowden syndrome. It does not replace regular medical care or discussions with your health care team. If you see an unfamiliar term, please see the Glossary of Terms.
That's not the name my doctor gave me
Cowden syndrome (CS)–first described in 1963–was named after Rachel Cowden, the young woman who had the features reported. However, Cowden syndrome is not the only name used for this condition. It is also known as the PTEN hamartoma syndrome (PHTS), or less commonly as the multiple hamartoma syndrome. Other related, but not identical, conditions include Bannayan-Ruvalcaba-Riley syndrome, Ruvalcaba-Myhre syndrome, Riley-Smith syndrome, or Bannayan-Zonana syndrome.
The use of different names can be confusing. The problem started when several different groups of physicians and researchers began describing collections of features they observed in their patients. Each group believed that they were describing a new condition. Because CS consists of various features that occur at different times, or not at all, different people will show different features even though they have the same genetic condition. That was enough to make researchers at the time believe they were describing different conditions.
In 1996, the overlap of features in CS and Bannayan-Ruvalcaba-Riley syndrome (BRRS) was recognized and shortly after it was shown that CS and BRRS were caused by harmful changes (called mutations) in the same gene. Since then, many names have been added to the list of related syndromes. Below are several other names that you may encounter as well.
- Ruvalcaba-Myhre-Smith syndrome
- Bannayan syndrome
- PTEN Hamartoma Tumor syndrome
- Autism disorders with macrocephaly
- Proteus-like syndrome
What is Cowden syndrome?
Cowden syndrome is a hereditary condition which causes multiple types of benign tissue overgrowth (called hamartomas) and a risk of breast, thyroid, and uterine cancers. The most consistent features of CS are small flesh-colored bumps on the skin involving a hair follicle (trichilemmomas) and small wart-like growths (papillomatous papules) on the face, hands and mouth. CS is also often associated with a large head size (macrocephaly) and hamartomatous polyps of the small and large intestine. Thyroid adenomas, goiter, and nodules are also seen at increased frequency. Women are at increased risk for benign breast conditions, such as ductal hyperplasia, papillomatosis, fibrocystic breast disease, or fibroadenomas. Less commonly seen are benign fatty tumors (lipomas), blood vessel growths (hemangiomas), and other changes.
The breast, thyroid and uterus are the most common sites for cancer development in CS. About 30-50 percent of women with CS will develop breast cancer, often at a much younger age than average. This is compared to a risk of about 12 percent for women without CS. It is also common for women with CS to eventually develop cancer in both breasts. Only two men with CS have been reported with breast cancer, so it is not clear if men with CS have an increased risk for developing breast cancer. Studies have suggested that uterine (endometrial) cancer is also increased in CS, with a risk currently estimated at six to eight percent (compared to about 2.5 percent in women with average risk). Men and women with CS have about a three to 10 percent lifetime risk for developing thyroid cancer (compared to one percent for an average risk person). Recent data suggests that the risk for colon cancer may also be increased. Cancers of the skin, kidney, small bowel and others have been reported, but it is has not been proven if the risks for these cancers are increased for people with CS.
Although it is not certain at what age the features of Cowden syndrome begin to appear, we believe that more than 90 percent of people with CS have some features by the age of 20. However, people diagnosed with CS do not necessarily have all the same features. For example, most individuals with CS have some skin feature, up to 75 percent have a breast-related problem, and about 80 percent have a large head (macrocephaly), but they may have only some or none of the other features. Table 1 lists the features that have been associated with Cowden syndrome and the approximate percentage of people that develop each feature (incidence).
Common characteristics of Cowden syndrome
| Class | Feature | Incidence |
|---|---|---|
| Skin and mouth lesions | Trichilemmomas, acral keratoses, and papillomatous papules | 90-100% |
| Head | Macrocephaly | 80% |
| Development | Intellectual disability/developmental delay | 15-20% |
| Thyroid problems | Goiter, adenomas, nodules | 40-60% |
| Breast problems (females) | Fibroadenomas, fibrocystic disease | 40% |
| Gastrointestinal | Hamartomatous polyps, ganglioneuromas | 80% |
| Gynecologic problems | Uterine fibroids (multiple and early onset) | 25% |
| Other benign growths | Lipomas Hemangiomas | 40-50% 10-40% |
| Cancer | Thyroid cancer Breast cancer Endometrial cancer | 3-10% 25-50% 6-8% |
What causes Cowden syndrome?
Cowden syndrome is a genetic syndrome usually caused by mutations in a gene known as PTEN. Mutations in this gene have been found in about 40-80 percent of people with a clinical diagnosis of CS and about half of all people with a clinical diagnosis of Bannayan-Ruvalcaba-Riley syndrome. The fact that both conditions are caused by mutations in the same gene explains why they share many similarities and why a physician must consider both possibilities when deciding upon a diagnosis and your health care management plan.
Genes serve as the basic unit of heredity, providing the body’s instructions for growth and development. Every cell of the human body has a complete set of this genetic information. Genes are composed of DNA and are housed on structures called chromosomes. There are 23 pairs of chromosomes (46 total). A child receives one chromosome of each pair from his or her mother's egg and one from their father's sperm. In this way, a child inherits half the information needed for development (including one copy of the PTEN gene) from each parent. Tests can be done to look for mutations in genes that can lead to disease.
The PTEN gene functions as a tumor suppressor. Tumor suppressor genes normally ensure that cells do not grow or divide more than they are supposed to. Only one copy of a tumor suppressor gene is needed to control cell growth. This means that when a person has inherited one PTEN gene with a mutation, the other, functional copy is still able to successfully control cell growth. However, if anything damages the second, functional copy of the PTEN gene in any cell, that person can develop either a benign and cancerous growth. Thus, although a person with CS inherits an increased risk for tumor development, they do not inherit the tumor or cancer itself.
How is Cowden syndrome diagnosed?
Physicians diagnose Cowden syndrome using a combination of criteria that were originally set by the International Cowden Syndrome Consortium and have been slightly revised over time. The various features are grouped into three categories: the pathognomonic criteria, the major criteria, and the minor criteria. Pathognomonic criteria are the most likely features to be associated with a condition. The major and minor criteria consist of related features that are not as specific to CS. The categories help to define when a patient really has CS instead of another condition that may have similar features. Your doctor, or a genetic specialist, can look at the features you have and compare them to the diagnostic criteria when deciding if CS is a likely possibility. An individual does not need to have all the features in every category to be diagnosed with CS, nor does someone necessarily have CS when they have only one feature from any or all of the categories. When making this diagnosis, it is more important to look at all of the features an individual experiences over time.
CS categories
- Pathognomonic criteria
- Mucocutaneous lesions
- Facial trichilemmomas
- Acral keratoses
- Papillomatous lesions
Major criteria
- Breast cancer
- Thyroid cancer
- Macrocephaly
- Lhermitte-Duclos disease
- Uterine (endometrial) cancer
Minor criteria
- Structural thyroid diseases (such as goiter or adenomas)
- Intellectual disability/developmental delay
- Gastrointestinal hamartomas or benign tumors
- Fibrocystic breast disease
- Lipomas
- Fibromas
How is Cowden syndrome inherited?
Cowden syndrome can be inherited or passed from an affected parent to a child. CS has an autosomal dominant pattern of inheritance. This means that each child (male or female) with an affected parent has a 50 percent chance of inheriting the PTEN gene mutation and developing CS. Likewise each child has a 50 percent chance of not inheriting the mutation and not developing CS (see diagram 1).
An evaluation of your family history can determine if a pattern exists. A family tree or a pedigree is a diagram of the members of your extended family, showing your family history.
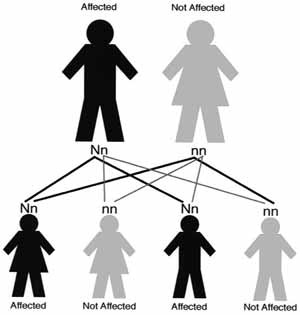
Diagram 1. This diagram shows how one gene from each parent is passed to a child. In the case above the father has one working gene, indicated by the lower case letter n, and one gene with a mutation, indicated by the capital letter N. The mother has two copies of normal genes. There are four possibilities of gene combinations for the child. Each child has a 50 percent chance of inheriting the mutation (N), regardless of the sex of the child.
Below is a family tree that demonstrates the pattern of autosomal dominant inheritance.
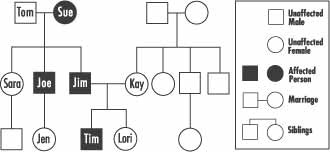
This family tree tells us several things about this family.
- Sue and Tom have three children, two of whom are affected. Since Sue was affected, each of her children had a 50 percent chance of inheriting her PTEN mutation and developing CS; Joe and Jim inherited the mutation.
- Sara did not inherit the mutation and does not have CS. She cannot pass this mutation on to her children, nor will they develop CS.
- Joe has one daughter who had a 50 percent chance of inheriting the mutation and developing CS. Jen did not inherit the mutation, will not develop CS, and cannot pass it to her children.
- Kay and Jim have two children. Tim inherited the mutation and he now has a 50 percent chance of passing it on to his children. Lori did not inherit the mutation and can not pass it to her children.
New mutations
Sometimes individuals with Cowden syndrome inherit their PTEN mutation from one of their parents. However, some individuals with Cowden syndrome have a PTEN mutation that occurred in either the egg or the sperm from which they were conceived; this is called a “new mutation.” A new mutation is not the result of something that did or did not happen in pregnancy, nor can it be prevented. When someone has Cowden syndrome as a result of a new mutation, they usually will not have relatives with Cowden syndrome. However they can have affected children.
Are tests for Cowden syndrome available?
Genetic testing of the PTEN gene can identify if someone has a mutation causing Cowden syndrome. There is a charge for these services when provided by clinically approved laboratories. These charges may or may not be covered by insurance and should be discussed with your doctor or genetic counselor.
The usual test for CS is called gene sequencing. In sequencing, the individual components (“letters”) of the PTEN gene are examined in detail looking for a mutation. This test is very accurate, but only about 40-80 percent of people meeting the criteria for CS have an identifiable mutation. This means that 20-60 percent of people who meet the criteria for CS will not have an identifiable mutation and will have a normal test result even though they do have CS. This could be due to a limitation in the test, and some labs offer additional testing to detect rare mutations missed by sequencing. It may also be possible, although this has not been proven, that CS could be cause by a mutation in another gene that has not been identified yet.
PTEN testing may also be available to you through a research study. Unlike clinical testing, research-based testing may take many months or years, but it is usually done without any cost to you. It may also provide information and results that are not available on a clinical basis. You may want to consider these issues as you decide whether you wish to pursue either clinical or research-based testing. Your physician and genetic counselor can help you if you are interested in participating in a research study.
What should I be doing about this condition?
Recommendations for cancer screening for people with Cowden syndrome are updated and published each year by the National Comprehensive Cancer Network (NCCN). In many cases, screening can help manage benign growths and detect any cancer at an early stage, when it is best treated. The recommended screenings for people with CS are summarized in Table 2.
Because of the risk for breast cancer, women with CS should have increased breast cancer screening. Currently this includes performing monthly breast self-examination, getting a breast exam from your doctor every 6 months beginning at 25 years of age, and an annual mammogram and breast MRI beginning at 30 to 35 years of age (or 5-10 years before the earliest known breast cancer in the family; whichever comes first). For some women with dense breast tissue that can make it harder to detect breast cancers, preventative mastectomy may be an option. Women should also be aware of the signs and symptoms of possible uterine cancer, such as abnormal vaginal bleeding, pelvic pain, pain during intercourse and painful urination.
Both men and women should receive thyroid cancer screening that includes an ultrasound of the thyroid at age 18 and annual thyroid palpation (having a doctor feel the thyroid) after that. You might also consider having an annual ultrasound. Because thyroid nodules can be a common feature, an experienced endocrinologist should evaluate any thyroid nodules to determine if they require follow-up, such as a biopsy.
Skin cancer screenings should also be considered. Yearly visits to a dermatologist can help manage the CS skin findings and also screen for skin cancer.
There is some recent evidence that the risk for colon cancer may be increased in people with CS. Currently it is recommended that people with CS have colonoscopy every 10 years beginning at age 50 like anyone in the general population. However, these recommendations could change and it is important to ask your doctor or genetic counselor about the most current guidelines.
Check yourself: Are you getting the screening you need?
| Beginning Age | Recommended Screenings | Frequency |
|---|---|---|
| 18 | Thyroid palpation and ultrasound | Annual for palpation consider annually for ultrasound |
| 18 | Breast self-examination | Monthly |
| 18* | Comprehensive physical exam | Annually |
| 25* | Clinical breast examination | Every six months |
| 30-35* | Mammography Breast MRI | Annual Annual |
| 30-35 | Endometrial cancer screening | Consider annually |
| Adults | Dermatology | Consider annually |
| 50 | Colonoscopy | Every 10 years, if normal |
| Beginning Age | Recommended Screenings | Frequency |
|---|---|---|
| 18* | Thyroid palpation and ultrasound | Annual for palpation consider annually for ultrasound |
| 18* | Thyroid ultrasound and palpation | Annual for palpation and consider for ultrasound |
| 18* | Comprehensive physical exam | Annual |
| Adults | Dermatology | Consider annually |
| 50 | Colonoscopy | Every 10 years, if normal |
* or 5-10 years before the earliest related cancer in the family.
Resources and support groups
Because Cowden syndrome can be complicated to diagnose, it is important to address your questions and concerns with your physician and genetic counselor. Remember that no two people with this diagnosis will have exactly the same symptoms. Pay particular attention to physical changes and discuss them with your physician regularly. Living with any type of genetic condition can be a challenge, but living with CS is a challenge you can face with a good medical team as your partner.
Remember that different people deal with this diagnosis in different ways. For some the path may be long and difficult while others adjust in a shorter time with less difficulty. Just remember to give yourself time to acknowledge your feelings. The members of your health care team--physicians, genetic counselors, and nurses--serve as an excellent resource and you should discuss your situation with them. Another valuable resource can be other people who have been in similar situations. Support groups can connect people who are dealing with similar issues and bring them together in a supportive environment. Here are several groups that may benefit you or someone you know.
American Cancer Society 1599 Clifton Road NE Atlanta, GA 30329 800-ACS-2345 www.cancer.org
Genetic Alliance4301 Connecticut Avenue NW Suite 404
Washington, DC 20008-2369
202-966-5557
https://geneticalliance.org
National Society of Genetic Counselors – NSGC 401 N. Michigan Avenue Chicago, IL 60611 312-321-6834 www.nsgc.org (includes a search tool to find a genetic counselor in your area)
Patient-run support groups*:
* These sites are for personal support for individuals and families with Cowden syndrome and are not monitored by medical professionals.
Glossary of terms
Acral keratoses:
small callous (thickening of the skin) usually located on the extremities
Adenoma:
a type of benign tumor in which the cells form glands
Angiolipomas:
benign growth of fat cells found in blood vessels
Baseline:
normal background level used for comparisons
Benign:
term describing a growth that is not cancerous
Benign breast disease:
a variety of conditions characterized by benign growths in breast tissue. See fibrocystic breast disease, ductal hyperplasia, intraductal papillomatosis, fibrocystic breast disease, and fibroadenoma
Chromosomes:
the structures made of DNA chains that contain genes
Colonoscopy:
examination of the colon using a long flexible scope (camera)
Cutaneous:
relating to the skin
DNA:
the genetic information that cells need for growth and development
Ductal hyperplasia:
overgrowth of cells lining the ducts in the breast. This is a common change in fibrocystic disease. Changes can range in severity from a benign growth to a pre-malignant condition that is associated with an increased risk for cancer development
Endometrium:
cells that line the inside of the uterus
Fibroadenomas:
a common benign growth that can occur in many different organs
Fibrocystic breast disease:
condition where the breast has lumpy and cystic fibrous tissue that is not due to tumor development.
Fibromas:
benign growth made of fibrous or fully developed connective tissue
Ganglioneuroma
: a benign tumor composed of nerve (ganglion) cells.
Gastrointestinal:
relating to the stomach and intestine
Gene:
the smallest units of heredity
Goiter:
enlargement of the thyroid gland.
Hamartoma:
benign growth of disorganized cells from normal tissue that stops itself from growing once the cells have reached maturity
Hamartomatous polyp:
a benign growth of disorganized cells from normal tissue that is found on a mucosal membrane (ie: inside lining of colon, mouth, nose, etc.). These polyps have a low chance of being malignant
Hemangioma:
common benign growth developed from blood vessels
Hereditary:
the genetic transmission of a particular trait from parent to child
Hyperextensibility:
extension of a part of the body beyond the usual limit
Hypotonia:
decreased or low muscle tone
Lhermitte-duclos disease (LDD): a benign tumor (called a dysplastic gangliocytoma)
in the part of the brain known as the cerebellum (found at the back of the head toward the base of the skull). LDD is believed to be a hamartomatous growth
Lipoma:
benign tumor made of fat tissue
Malignant:
cancerous growth that tends to become progressively worse and often invades other tissues.
Mammograms:
low-dose x-ray that can detect breast cancer up to two years before a woman or a health professional can feel any signs of it
Macrocephaly:
larger head size
Mucocutaneous:
affecting the skin and inside of the mouth
Myopathic:
a muscle problem
Pectus excavatum:
depression of the breastbone and rib cartilage.
Papillomatous:
multiple benign growth arising from specific types of cells (epithelial cells) which line many organs and tissues in the body.
Pathognomonic criteria:
characteristic or indicative of a disease, i.e. one or more symptoms, findings, or pattern of findings specific for a given condition that are usually not found in any other condition
Pedigree:
pictorial relation of yourself and your family history
Palpation:
light touch by a clinician to feel structures under the skin
Papillomatous lesions:
benign growth that is wart-like (but are not warts)
Prophylactic tissue and organ removal (mastectomy):
surgical removal of tissue or organ used as a method of risk reduction for cancer
PTEN:
tumor suppressor gene located on chromosome 10 that when mutated can cause Cowden syndrome and other conditions.
Scoliosis:
curvature of the spine
Sequencing:
a detailed laboratory test that can detect very small changes in a gene
Syndrome:
set of features that occur together
Trichilemmomas:
hamartoma of the lower outer root and sheath of the hair root.
Tumor suppressor gene:
a gene whose function is to monitor and control cell growth
Ultrasound:
technique that uses sound waves to visualize internal structures of the body
References
- Eng C. Will the real Cowden syndrome please stand up: revised diagnostic criteria. J Med Genet 2000: 37(11): 828-30.
- Heald, B, Mester, J., Rybicki, L., Orloff, M.S., Burke, C.A, and Eng, C. Frequent Gastrointestinal Polyps and Colorectal Adenocarcinomas in Prospective Series of PTEN Mutation Carriers. Gastroenterology, epub 27 Jun 2010.
- National Comprehensive Cancer Network. Clinical Practice Guidelines in Oncology - Genetic/Familial High-Risk Assessment: Breast and Ovarian. http://www.nccn.org/professionals/physician_gls/PDF/genetics_screening.pdf
- Nelen, M. R., G. W. Padberg, et al. (1996). "Localization of the gene for Cowden disease to 10q22-23." Nature Genetics 13: 114-116
- Pilarski, R. Cowden syndrome: a Critical Review of the Clinical Literature. J Genet Counsel 2009;18:13-27.
- Pilarski, R., Stephens, J., Noss, R., Fisher J.L. and Prior, T (2011). Predicting PTEN mutations: an evaluation of Cowden syndrome and Bannayan-Ruvalcaba-Riley syndrome clinical features.
Credits
2010 revisions by:
- Joy Larsen Haidle, MS, CGC, Genetic Counselor, Humphrey Cancer Center
- Heather Hampel, MS, CGC, Genetic Counselor, Clinical Cancer Genetics Program and Division of Human Genetics, The Ohio State University
- Robert Pilarski, MS, CGC, MSW, LSW, Genetic Counselor, Clinical Cancer Genetics Program and Division of Human Genetics, The Ohio State University
Original version written by:
- Sarah Burton, University of Iowa Health Care
- Joy Larsen Haidle, MS, CGC
- Heather Hampel, MS, CGC
- Charis Eng, MD, PhD
Illustrations by Terry Demmer
Special Thanks to: University of Iowa Health Care, Division of Medical Genetics