UI Health Care begins first employee COVID-19 vaccinations with the vaccine it tested in clinical trials
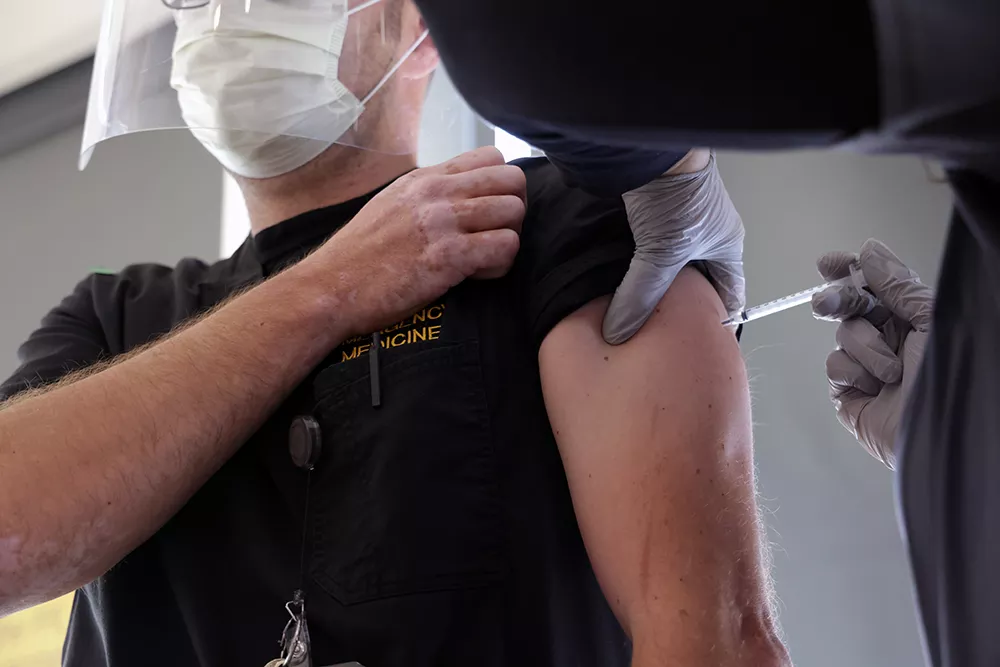
Today, University of Iowa Health Care is the first in the state to start vaccinating employees with the Pfizer-BioNTech Vaccine (Comirnaty) against COVID-19. The vaccine was approved for emergency use by the Food and Drug Administration (FDA) on Friday, Dec. 11 and is prioritized by the Centers for Disease Control and Prevention (CDC) for health care workers and residents of long-term care facilities.
“This is an historic moment to change the course of the pandemic and we are proud of our role as leaders in this process,” says Suresh Gunasekaran, MBA, chief executive officer of UI Hospitals & Clinics and associate vice president of UI Health Care.
During the first day of vaccinations, 178 UI Health Care employees received the first of two doses of the vaccine.
Because of the limited number of doses initially available and allocated to UI Health Care, the first group of employees to be vaccinated are primarily front-line providers and staff working in the units that directly care for patients with COVID-19. Several essential leaders to the COVID-19 pandemic response will also lead the way in being among the first vaccinated.
These first employees represent the majority of UI Health Care who have indicated their intent to be vaccinated. Based on over 12,000 responses to an internal survey of UI Health Care employees last week, 85% indicated that they are willing to receive the vaccine when they are offered it.
View photos from the Pfizer-BioNTech Vaccine (Comirnaty) delivery in our gallery. It arrived and was distributed to the first round of UI Health Care employees earlier today.
UI Health Care’s role in helping develop the Pfizer-BioNTech Vaccine (Comirnaty)
Patricia Winokur, MD, the executive dean of the Carver College of Medicine and the principal investigator for University of Iowa’s Pfizer clinical trial site, has long-standing expertise in running vaccine clinical trials and is confident in this vaccine’s ability to safely provide protection against COVID-19.
“Having coordinated our part of this clinical trial and seen first-hand how our participants have fared, I can assure you that all the data tells us that this is a highly effective and safe vaccine,” Winokur says. “When my turn comes to receive the vaccine, I will absolutely be rolling up my sleeve to get it.”
UI Health Care’s role doesn’t stop there when it comes to how this historic moment in the pandemic came to light so quickly in terms of vaccine development and its approval for emergency use.
Stanley Perlman, MD, PhD, a UI professor of microbiology and immunology, and pediatrics, has been studying coronaviruses for almost 40 years and has shared his expertise with numerous national and international committees. He has informed vaccine development, was part of the international committee that named the virus SARS-CoV-2, and his most recent involvement was as an advisor to the FDA on approval of the vaccine.

Patience required as the production and approval of COVID-19 vaccines continue
As more vaccines are approved and more vaccine doses are manufactured and distributed, the CDC will open access to and prioritization for other population groups, including older adults and people with underlying health risks.
UI Health Care is charged first with vaccinating its employees and looks forward to more information on when the vaccines will become available to more of the general public.
It’s necessary to keep up safety measures
It will take time for everyone who wants to be vaccinated to have the required doses, so until then all Iowans should please continue to take the steps necessary to protect themselves, their families, and the most vulnerable within our communities. Everyone should continue to wear a face mask, avoid large gatherings, maintain social distance, and wash their hands frequently.
Get the latest on UI Health Care's COVID-19 response